Fluorescence Microscopy
The fluorescence microscopy developed to one of the most important tools of the life science area. The observation of the fluorescence emergences in biological samples delivers differentiates conclusions about morphology and functionality of cells. The objects of examination reach from fixated cells via chromosomes and genes up to observation of processes in living cells.
Due to new techniques and applications, like the fluorescence angiography for examination of ocular fundus, the fluorescence microscopy has blazed a trail into the field of medical diagnostics and therapy. Special importance is set to the possibilities of the intra-operational use.
As highly sensitive and selective tools techniques of fluorescence endoscopy are available in cancer diagnostics. Main topics are here molecular imaging and optical biopsy.
What is Fluorescence?
The term fluorescence describes a short-time spontaneous emission of light during the change of an electrically excited status to a state of low energy. As with phosphorescence, it is about a kind of luminescence. The difference of fluorescence and phosphorescence lies in the considerably shorter lighting period of the fluorescence. The process of fluorescence cannot be traced back to a thermal glowing or reflection.

Figure 1. : Excitation and Emission
The change from an energetically low state to and energetically high state can be performed by exposure with light of a certain wave length. This results in absorption of a Photon. Returning to an energetically lower state the emission of a photon follows – the fluorescence photon.
By different processes leading back to the energetically initial state the wave length of the emitting light is longer, respectively energetically lower opposite to the exciting light. The difference between excitation and emission spectrum is called Stokes Shift.
 |
By adjustment of excitation and emission light the spectrums can be separated by respective filter. The knowledge of excitation and emission wave length is important especially for the development and design of fluorescence microscopes. |
Fluorochromes
With observation of fluorescence one differentiates between primary and secondary fluorescence. Primary fluorescence means the natural emission of fluorescence photons without additional preparation steps. Molecules, which do not show the property of primary fluorescence, can be used in a fluorescence examination by being marked with a fluorochrome. Such a marking is called fluorochromation or secondary fluorescence.
There are different types of microscopes available for observation of fluorescence samples. By means of a wide field microscope biological processes can be observed. The confocal laser-scanning microscopy and multi-photon microscopy offer the possibility to obtain a significantly higher local resolution as well as z-stacking of the sample. With these techniques the scattered light appearing with wide field microscopes can be minimized or even totally avoided, as the fluorescence excitation only happens within the focus plane.
Cameras for Fluorescence Imaging
Spectrum: when you select a camera make sure the camera spectral sensitivity range covers the range of wavelengths you are dealing with. This becomes matter when you deal with InfraRed (IR) or above since regular cameras work mainly for the Visible range (400-700nm).
Camera Gain:
When working with objects under low light conditions (e.g. in dark field or fluorescence applications), the image signal can be amplified by using the “Gain“-slider control. This allows a reduction of the exposure time in order to achieve a maximum image frame rate of the live image. Please note that the use of this function results in a simultaneous increase of the background noise in the image.
Software for Fluorescence Image Acquisition
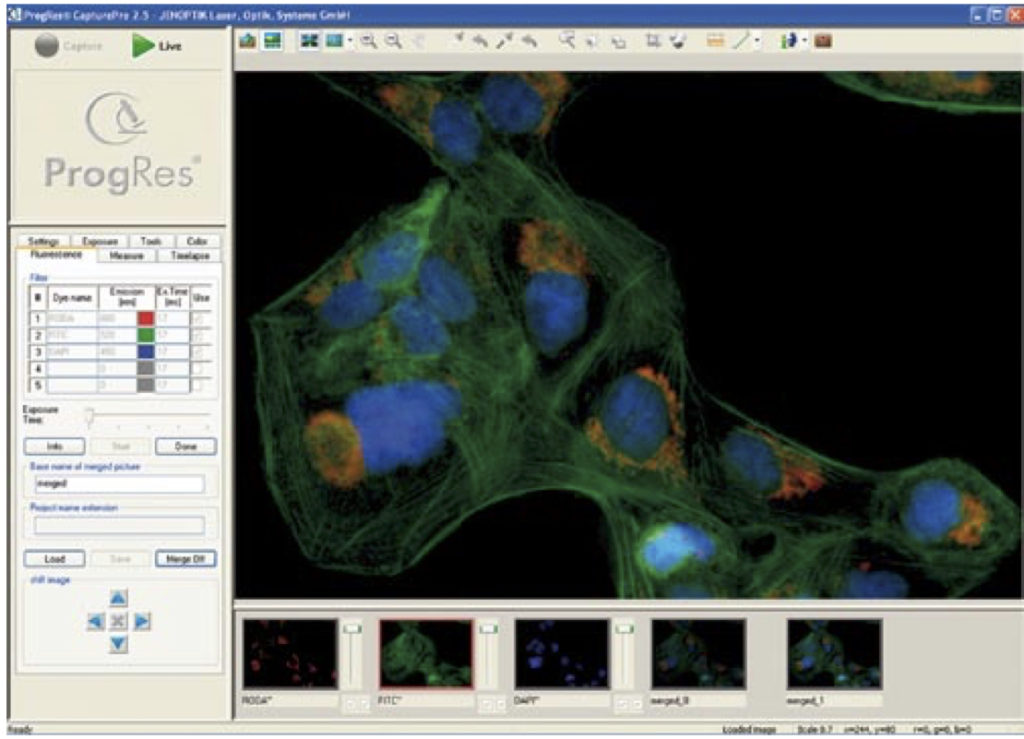
Some camera softwares, either free license or paid,offers a unique fluorescence mode for easy capturing and merging of monochrome and bright-field images.ProgRes CapturePro is one of the free softwares (which works on Jenoptik Cameras only). With the functions in the “Fluorescence” register tab, images of single or multi-color fluorescence can be captured.
 |
Some software offers the possibility to capture several single monochrome images, color them automatically and merge to one final multi-color fluorescence micrograph. In this example, you can enter up to 5 different filters or fluorochromes in the column “Dye name”. Afterward, enter the corresponding emission value of the dye in the fields of the Emission” column. In the column, Ex.Time (ms) the exposure time can be manually inserted before the fluorescence measurement is carried out. Alternatively, it can be inserted after the start of the fluorescence capturing. |
Particular applications of fluorescence microscopy
Fluorescence in-situ Hybridisation (FISH)
- direct, simple and fast localisation of nucleon acid sequences
- use of a marked probe, being hybridised over base pairs to the nucleon acid to be proved
- characterized by high sensitivity and specifity
- technique uses the re-association kinetics of double strand nucleon acid molecules
- after hybridization of the probe to the target sequence the fluorescence signal of the market probe can be detected and hence the demanded sequence located
- by means of FISH pre- and post natal chromosome analysis can be performed ( analysis of chromosome structure, appraisal of numerical or structural mutations)
Fluorescence Lifetime Imaging Microscopy (FLIM)
- measure of fluorescence live time as additional value for examination of biological processes
- besides the knowledge about the morphology, exceeding detailed information about environmental
- temperature of the fluorophors can be obtained
- measurement in time- and phase-regions (Frequency Domain) are possible
- time-resolved life time measurements: intensity integrating measurements, Time Correlated Single Photon Counting (TCSPC), gated fluorescence detection
Fluorescence Resonance Energy Transfer (FRET)
- firstly in 1948 described by Th. Foerster, therefore often called Foerster Resonance Energy Transfer
- principle: see above
- can be detected by reduction of donor fluorescence or increase of acceptor fluorescence
- being used a optical nano-measure, as FRET only appears in-between a certain molecule distance (max. 5nm)
- further being used to visualise timely and spacious changes within a cell
- proof of protein-protein, protein-nucleon acid, and nucleon acid-nucleon acid – inter actions
- FRET signal alternates corresponding to the distance of the marked molecules
Fluorescence Recovery After Photo-bleaching (FRAP)
- possible method to testify transport processes within a cell
- determination of concentration balance processes as well as the time-space resolved detection of molecule movements as a function within cells, which is – besides the morphological determination of single components – the second main aspect of fluorescence microscopy
- with FRAP the fluorescence dye is being destroyed by intensive radiation of a region of the sample
- the returning period of the fluorescence provides information about the flexibility and speed of the fluorescence dye within the object
Sample Images
Captured using our IncuScope with 3 LEDs (UV, Blue and Green), 3MP sCMOS camera BIC-C30, and 20x objective lenses, using IncuView software. Sample: FluoCells® Prepared Slide #2: bovine pulmonary artery endothelial cells (BPAE) cells with Mouse Anti-α-tubulin, BODIPY FL Goat Anti-Mouse IgG, Texas Red-X Phalloidin, and DAPI), (Molecular Probes) Further details: https://lnkd.in/efqpv2E


That’s good to know that you could get a bunch of different colors using that they of the microscope. I would think that would be a good way to make sure that you could see the individual parts of the object you are looking at. I would also think that it would be useful to make it stand out from the background.